Executive Neural Brief
Dopamine is a catecholamine neurotransmitter functioning as the primary chemical messenger for incentive salience, executive function, and motor control within the central nervous system. While often colloquially reduced to a “pleasure molecule,” its neurophysiological role is far more complex, acting as a modulator of motivation rather than a direct generator of hedonic experience. This hub examines the neurophysiology of incentive salience through three primary mechanisms: dopaminergic signaling dynamics, mesolimbic pathway function, and the Real-Time Neuroplasticity™ framework for motivation optimization.
Core Neurobiological Mechanisms
Dopaminergic Signaling Dynamics: Motivation relies on the interplay between tonic (baseline) firing rates and phasic (burst) release events. Phasic dopamine release acts as a teaching signal through Reward Prediction Error (RPE), reinforcing synaptic connections when outcomes exceed expectations. Higher motivation paradoxically links to lower reward-evoked dopamine release, as anticipated rewards generate smaller prediction errors than unexpected ones.
Mesolimbic Pathway Function: The ventral tegmental area (VTA) to nucleus accumbens (NAc) circuit is responsible for “incentive salience”—transforming neutral stimuli into attractive targets commanding attention and approach behavior. This pathway processes the raw “value” of stimuli, determining whether potential rewards justify energy expenditure. Dysregulation here is central to motivation disorders, with hypoactivity linked to depression’s avolition and hyperactivity associated with addiction’s compulsive “wanting.”
Modern Environmental Mismatch: The dopaminergic system evolved for naturalistic stimuli requiring metabolic expenditure, but modern inputs (addictive substances, hyper-stimulating activities) trigger supraphysiological surges without corresponding effort. This creates a “short circuit” in cost-benefit analysis, leading to receptor downregulation, blunted reward sensitivity, and altered temporal discounting that collapses long-term motivation.
Clinical Implications
Dopamine-driven motivation represents not a simple pleasure response but a sophisticated neurochemical calculation of “return on investment” (ROI) between metabolic cost and predicted survival value. Modern dysregulation manifests as progressive desensitization where natural rewards generate insufficient dopamine to motivate action, locking individuals in states of anhedonia where only supranormal stimulation generates motivational salience.
 Figure: Synaptic constellation visualization of dopamine timing in motivation
Figure: Synaptic constellation visualization of dopamine timing in motivation
The Neurophysiology of Incentive Salience: Dopaminergic Mechanisms
Dopamine is a catecholamine neurotransmitter functioning as the primary chemical messenger for incentive salience, executive function, and motor control within the central nervous system. While often colloquially reduced to a “pleasure molecule,” its neurophysiological role is far more complex, acting as a modulator of motivation rather than a direct generator of hedonic experience. It reinforces behaviors essential for survival—such as caloric acquisition and shelter seeking—by encoding the perceived value of an action relative to its metabolic cost. This monoamine is synthesized in specific midbrain nuclei and projected across distinct pathways to regulate focus, working memory, and the drive to exert effort.
The fundamental mechanism of dopamine-driven motivation relies on the interplay between tonic (baseline) firing rates and phasic (burst) release events. Phasic dopamine release acts as a teaching signal, reinforcing synaptic connections through long-term potentiation (LTP) when an outcome exceeds expectations. This process, known as Reward Prediction Error (RPE), is the neurobiological basis of learning and motivation. When a behavior results in a beneficial outcome, the resultant dopaminergic surge strengthens the neural circuits responsible for that action, increasing the probability of repetition. Conversely, the absence of an expected reward depresses these circuits, extinguishing the behavior.
Homeostatic Dysregulation and Receptor Downregulation
The dopaminergic system is evolutionarily calibrated for naturalistic stimuli, such as food or social bonding, which produce moderate, manageable increases in synaptic dopamine. However, modern environmental inputs—specifically addictive substances and hyper-stimulating activities like gambling—trigger supraphysiological surges of dopamine in the synaptic cleft. These exaggerated spikes overwhelm the post-synaptic neurons, forcing the brain to engage neuroprotective homeostatic mechanisms to prevent excitotoxicity.
The primary mechanism of this adaptation is the downregulation of dopamine receptors, specifically the D2 receptor subtype. In response to chronic overstimulation, post-synaptic neurons internalize these receptors, effectively reducing the density of binding sites available for neurotransmission. Simultaneously, the presynaptic neurons may reduce dopamine synthesis and release. This results in a blunted sensitivity to reward, a state clinically manifesting as anhedonia. Consequently, the individual experiences diminished pleasure from everyday activities that previously elicited a positive response. Motivation shifts from a pursuit of growth to a compulsive drive to restore dopamine levels to normalcy, sustaining the addiction cycle despite diminishing returns.
 Figure: Neural circuit visualization of dopamine prediction in motivation systems
Figure: Neural circuit visualization of dopamine prediction in motivation systems
Striatal Encoding: Reward Magnitude and Sunk Costs
Recent neurophysiological research has elucidated the nuanced role of striatal dopamine in cost-benefit analysis. The striatum does not merely signal the presence of a reward; it integrates complex variables including reward magnitude (the potential benefit) and sunk costs (the effort already expended). This integration is critical for decision-making processes that determine whether to persist in a task or abandon it.
A paradox exists within striatal kinetics regarding the relationship between motivation levels and dopamine release. While high tonic levels of dopamine generally correlate with high motivation and vigor, phasic release profiles tell a different story. Research indicates that higher motivation is paradoxically linked to lower reward-evoked dopamine release. This inverse correlation reconciles conflicting findings across temporal scales: when an organism is highly motivated and expects a reward, the eventual receipt of that reward generates a smaller prediction error (and thus a smaller phasic spike) because the outcome was anticipated. Conversely, unexpected rewards generate massive phasic spikes to update the brain’s internal model of value. This suggests that dopamine dynamics encode the precision of reward expectation as much as the reward itself.
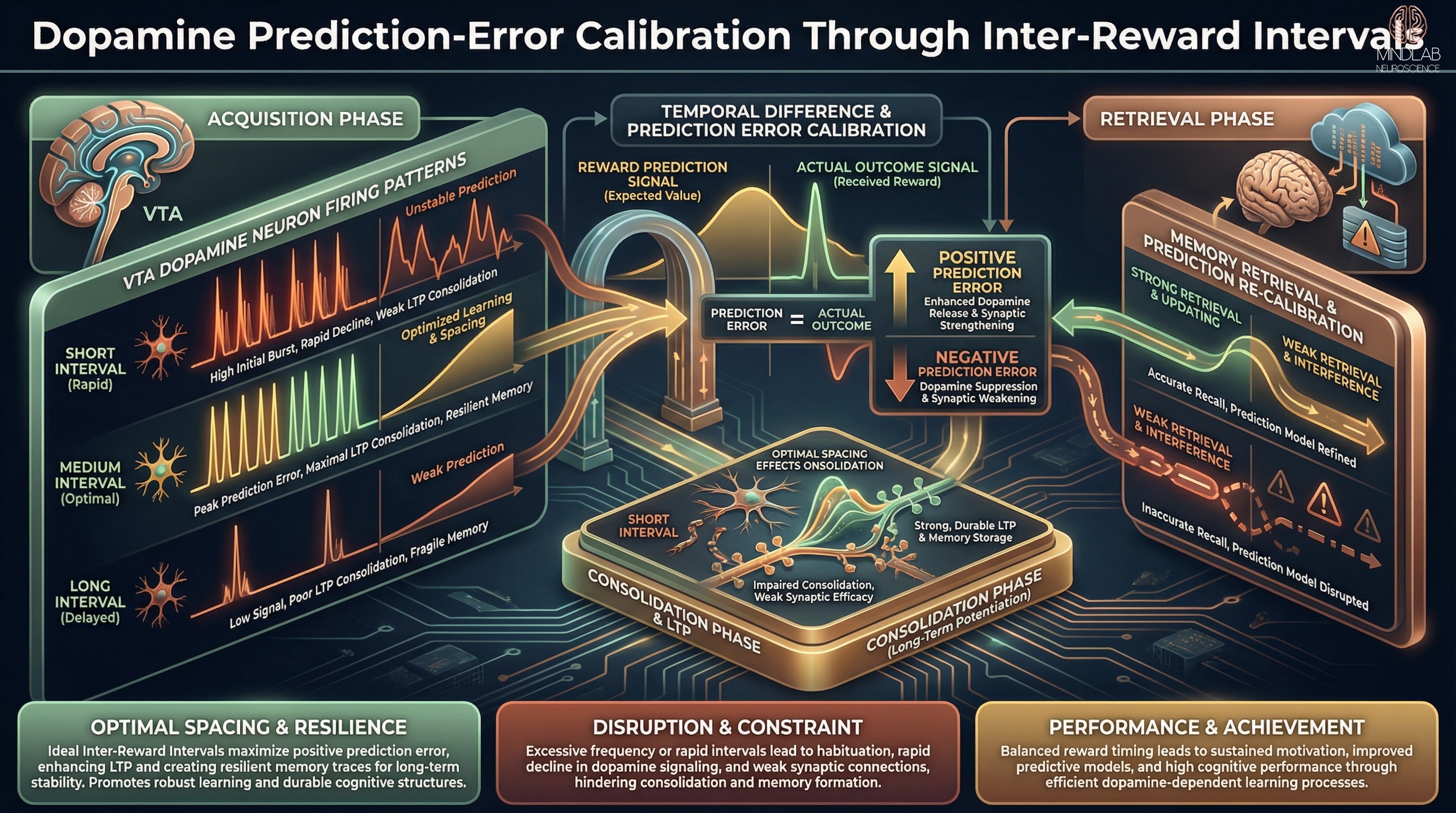 Figure: Diagrammatic visualization of dopamine prediction in motivation and learning
Figure: Diagrammatic visualization of dopamine prediction in motivation and learning
The Mesolimbic Pathway: The Engine of “Wanting”
The mesolimbic pathway is the anatomical core of the brain’s reward and motivation circuitry. Originating in the Ventral Tegmental Area (VTA) of the midbrain, dopaminergic neurons project to the Nucleus Accumbens (NAc) located in the ventral striatum. This circuit is responsible for “incentive salience”—the attribute that transforms a neutral sensory stimulus into an attractive target that commands attention and induces approach behavior.
Activity within the VTA-NAc circuit fluctuates to influence the composition of naturalistic behaviors. It processes the raw “value” of a stimulus, determining whether the potential reward justifies the expenditure of energy. When the VTA fires, it floods the NAc with dopamine, disinhibiting motor pathways and facilitating the initiation of action sequences. Dysregulation here is central to disorders of motivation; hypoactivity is linked to the avolition seen in depression, while hyperactivity is associated with the compulsive “wanting” characteristic of addiction, distinct from the “liking” (hedonic impact) mediated by opioid systems.
The Nigrostriatal Pathway: Motor Control and Effort Calculation
Parallel to the mesolimbic system, the nigrostriatal pathway projects from the Substantia Nigra pars compacta (SNc) to the Dorsal Striatum (specifically the dorsolateral striatum). While traditionally associated with motor control and the pathophysiology of Parkinson’s disease, this pathway is integral to the motivational aspect of effort exertion. It serves as the bridge between the “will” to act and the “execution” of the movement.
The nigrostriatal system incorporates “effort costs” into the motivational equation. Dopamine release in the dorsal striatum inversely correlates with the perceived difficulty of a task. When dopamine levels in this pathway are depleted, organisms become “effort averse,” choosing low-reward/low-effort options over high-reward/high-effort options. This pathway ensures that motivation is not merely an abstract desire but is physiologically coupled with the motor resources required to achieve the goal. In addiction, the transition from recreational use to compulsive habit involves a shift in control from the ventral striatum (mesolimbic) to the dorsal striatum (nigrostriatal), cementing the behavior as an automatic motor routine.
Neuromodulatory Interactions
Dopamine does not operate in a vacuum; its efficacy is modulated by interactions with other neurotransmitter systems. Acetylcholine plays a supporting role in the striatum, balancing dopaminergic signaling to regulate learning and flexibility. Cholinergic interneurons modulate dopamine release, influencing how the brain transitions between established behaviors and new learning opportunities.
Furthermore, the Locus Coeruleus, the brain’s primary source of noradrenaline (norepinephrine), projects to dopaminergic centers. Noradrenergic modulation enhances synaptic plasticity and arousal, sharpening the signal-to-noise ratio of dopaminergic firing. This interaction ties motivational states to physiological arousal, ensuring that the organism is not only motivated to act but is metabolically prepared to execute the necessary behaviors. These conserved evolutionary pathways highlight that motivation is a systemic physiological state, integrating sensory prediction, metabolic cost calculation, and motor readiness.
The Phylogeny of Drive: Dopamine as a Foraging Algorithm
To understand the dysregulation of motivation in the modern clinical context, we must first deconstruct the evolutionary architecture of the mesolimbic dopaminergic system. Contrary to popular reductionist views, dopamine is not a hedonic generator; it is a functional algorithm designed for resource acquisition. In the Pleistocene environment, survival was predicated on the organism’s ability to overcome the energetic cost of movement to secure scarce resources.
The brain, comprising roughly 2% of body mass but consuming 20% of total glucose, operates under strict metabolic constraints. Consequently, the release of dopamine from the ventral tegmental area (VTA) to the nucleus accumbens (NAc) serves as a neurochemical calculation of “return on investment” (ROI). It bridges the gap between the metabolic cost of action and the predicted survival value of the reward.
In the ancestral context, this algorithm was calibrated for uncertainty. Foraging for food involved traversing variable terrain, expending calories with no guarantee of reward. Dopamine was released not at the moment of consumption but during the pursuit phase—the act of searching, tracking, and securing the resource. This anticipatory spike motivated persistence despite energetic costs. The dopaminergic burst encoded the effort-to-reward ratio, ensuring that organisms only pursued targets with a favorable metabolic payoff.
The Modern Displacement: Hijacking the Foraging Circuit
The modern environment has introduced a critical mismatch between the ancestral calibration of dopamine signaling and the structure of contemporary reward systems. In the Pleistocene, effort and reward were tightly coupled in time and space. The hunt required physical exertion, spatial navigation, and social coordination, all of which terminated in the tangible reward of caloric intake. The dopamine system evolved to reinforce this cycle.
Today, however, this circuit is exploited by stimuli that deliver dopamine spikes with minimal effort. Social media notifications, processed foods, and pornography provide immediate, supranormal dopamine release without the metabolic expenditure the system was designed to measure. This creates a “short circuit” in the cost-benefit analysis. The brain receives the neurochemical signal of a successful hunt but without the corresponding caloric expenditure or adaptive value.
The consequence is a progressive desensitization of the dopaminergic system. As the baseline level of stimulation increases, natural rewards—such as meaningful work, physical exercise, or social bonding—generate insufficient dopamine release to motivate action. The individual becomes locked in a state of anhedonia, where the only activities capable of generating motivational salience are those that deliver supranormal stimulation. This is the mechanistic basis of behavioral addiction.
Temporal Discounting and the Collapse of Long-Term Motivation
One of the most devastating consequences of modern dopaminergic dysregulation is the alteration of temporal discounting—the degree to which the brain devalues future rewards in favor of immediate ones. In a neurologically optimized state, the prefrontal cortex (PFC) exerts top-down control over the mesolimbic dopamine system, allowing the individual to delay gratification in pursuit of larger, more meaningful goals.
However, chronic exposure to immediate, high-dopamine stimuli weakens the structural connectivity between the PFC and the nucleus accumbens. Functional MRI studies reveal that individuals with addictive behaviors exhibit reduced gray matter volume in the PFC and diminished functional connectivity to subcortical reward centers. This structural degradation impairs the brain’s ability to engage in prospective thinking—the simulation of future states necessary for long-term planning.
Without this capacity, motivation collapses into a series of impulsive reactions to immediate stimuli. The individual loses the ability to sustain effort toward distant goals, as the dopaminergic system is no longer responsive to delayed rewards. This is not a failure of willpower; it is the predictable outcome of a neural circuit operating under conditions for which it was not designed. The brain has been hijacked by an environment that exploits its most ancient vulnerabilities.
Mechanisms of Real-Time Neuroplasticity™
Long-Term Potentiation: Strengthening Desired Circuits
Long-Term Potentiation (LTP) is the process by which repeated co-activation of two neurons increases the efficiency of their connection. When a pre-synaptic neuron fires simultaneously with a post-synaptic neuron, the synapse between them is strengthened through the insertion of additional AMPA receptors into the post-synaptic membrane. This increases the post-synaptic neuron’s sensitivity to glutamate, the brain’s primary excitatory neurotransmitter.
The initiation of LTP is gated by attention. When the brain is in a state of focused attention, the nucleus basalis releases acetylcholine (ACh) into the cortex. ACh acts as a “tagging” molecule, marking the synapses that are active during the focused state for strengthening. This is the neurobiological basis of “deliberate practice.” Simply repeating a task without focused attention does not induce LTP; the neural substrate is not tagged for modification.
In the context of Real-Time Neuroplasticity™, LTP is the mechanism by which desired dopaminergic pathways are reinforced. By engaging in tasks that require sustained focus and produce moderate dopamine release, the brain strengthens the neural circuits associated with productive behaviors. This process is not passive; it requires the strategic application of attention to ensure that the correct pathways are tagged for strengthening.
Long-Term Depression and Synaptic Pruning: Eliminating Competing Pathways
While LTP strengthens desired circuits, Long-Term Depression (LTD) weakens and eliminates the maladaptive ones. LTD occurs when a synapse is repeatedly activated out of synchrony with the post-synaptic neuron. This desynchronization triggers the removal of AMPA receptors from the post-synaptic membrane, reducing the strength of the connection.
Over time, weakened synapses are physically eliminated by microglia—the brain’s resident immune cells. Microglia continuously survey the brain, identifying and pruning synapses that have been marked for removal by LTD. This process is not merely passive degradation; it is an active remodeling of the neural architecture.
In the Real-Time Neuroplasticity™ framework, LTD is the mechanism by which addictive or maladaptive dopaminergic pathways are dismantled. By deliberately avoiding stimuli that trigger supranormal dopamine release and instead engaging in activities that produce moderate, sustained dopamine elevations, the brain progressively weakens the neural circuits associated with addictive behaviors. This is the biological basis of “extinction” in addiction treatment.
Myelination: Bandwidth and Synchronization
The final component of the RTN mechanism is the structural reinforcement of the signal pathway, known as myelination. Myelin is the fatty sheath of insulation that wraps around the axons of neurons, produced by glial cells called oligodendrocytes.
While synaptic changes (LTP/LTD) can happen in seconds to minutes, myelination is the process that solidifies these changes for high-speed performance. Myelin serves two distinct functions in this framework:
Velocity: It increases the speed of electrical transmission (action potentials) by up to 100 times.
Refractory Reset: It reduces the recovery time required for a neuron to fire again, allowing for higher frequency signal bursting.
Crucially, recent research indicates that electrical activity in axons can stimulate oligodendrocyte precursor cells (OPCs) to begin differentiating and wrapping axons almost immediately. Within the RTN paradigm, the goal is to trigger this proliferation through high-fidelity signal repetition. By synchronizing the firing of distant brain regions (e.g., the prefrontal cortex and the cerebellum), we induce a demand for tighter timing. The brain responds by insulating the connecting fibers to ensure that signals arrive exactly when needed.
Synthesis: The RTN Loop
These three mechanisms do not operate in isolation; they are synergistic forces within the Real-Time Neuroplasticity™ ecosystem.
Directed Neuroplasticity highlights the desired circuit through intense focus (Acetylcholine).
Synaptic Pruning suppresses the competing, inefficient circuits (LTD/Microglia).
Myelination locks in the optimal path, transforming a conscious effort into an unconscious, high-speed reflex (Oligodendrocytes).
Frequently Asked Questions
What is the fundamental difference between dopamine’s role in “wanting” versus “liking”?
Dopamine is primarily responsible for “incentive salience” or “wanting”—the motivational drive to pursue a reward. This is distinct from “liking” or the hedonic pleasure of consumption, which is mediated by opioid systems in the brain. The mesolimbic dopamine pathway (VTA to nucleus accumbens) generates the desire to act, while separate circuits involving the ventral pallidum and orbitofrontal cortex generate the pleasure of reward consumption. This distinction explains why individuals with addiction continue to compulsively “want” substances even when they no longer “like” them.
How does chronic stress affect dopamine signaling and motivation?
Chronic stress induces profound alterations in dopaminergic function through several mechanisms. Elevated cortisol levels downregulate dopamine receptors in the prefrontal cortex and striatum, reducing sensitivity to reward. Stress also increases dopamine turnover in the mesolimbic pathway, creating a state of hyperarousal that paradoxically diminishes motivation for non-stress-related goals. This neurochemical shift prioritizes survival-oriented behaviors at the expense of long-term planning and goal-directed action, contributing to the motivational deficits observed in stress-related disorders.
What is “reward prediction error” and why is it central to dopamine function?
Reward Prediction Error (RPE) is the discrepancy between expected and actual reward outcomes, serving as the primary teaching signal in dopaminergic circuits. When an outcome exceeds expectations, phasic dopamine bursts reinforce the neural pathways responsible for the successful behavior. When outcomes fall short of expectations, dopamine firing decreases, weakening those pathways. This computational mechanism allows the brain to continuously update its internal model of value, optimizing decision-making and motivation based on environmental feedback rather than fixed reward values.
Can dopamine sensitivity be restored after chronic overstimulation?
Yes, dopamine sensitivity can be restored through strategic neuroplastic interventions. The Real-Time Neuroplasticity™ framework employs three mechanisms: (1) Long-Term Depression to weaken maladaptive pathways through deliberate avoidance of supranormal stimuli, (2) Long-Term Potentiation to strengthen natural reward circuits through focused engagement with moderate-challenge activities, and (3) Myelination to solidify these changes into high-speed neural pathways. This process requires consistent application over weeks to months, as receptor upregulation and synaptic remodeling operate on biological timescales rather than immediate behavioral change.
What role does dopamine play in learning versus motivation?
Dopamine serves dual functions in learning and motivation through distinct temporal dynamics. For learning, phasic dopamine bursts following unexpected rewards (positive prediction errors) strengthen synaptic connections via long-term potentiation, encoding new associations between actions and outcomes. For motivation, tonic dopamine levels in the nucleus accumbens modulate the “vigor” or intensity of goal-directed behavior, determining how much effort an organism will expend for potential rewards. These functions are interconnected but separable—learning establishes what is valuable, while motivation determines whether to pursue that value given current energetic constraints.
 Figure: Organic topography visualization of dopamine timing mechanisms
Figure: Organic topography visualization of dopamine timing mechanisms
Selected Scientific References
- Schultz, W. (2016). Dopamine reward prediction error coding. Dialogues in Clinical Neuroscience, 18(1), 23–32.
- Berridge, K. C., & Robinson, T. E. (2016). Liking, wanting, and the incentive-sensitization theory of addiction. American Psychologist, 71(8), 670–679.
- Niv, Y., Daw, N. D., Joel, D., & Dayan, P. (2007). Tonic dopamine: opportunity costs and the control of response vigor. Psychopharmacology, 191(3), 507–520.
- Volkow, N. D., Wang, G. J., Fowler, J. S., & Tomasi, D. (2012). Addiction circuitry in the human brain. Annual Review of Pharmacology and Toxicology, 52, 321–336.
- Wise, R. A. (2004). Dopamine, learning and motivation. Nature Reviews Neuroscience, 5(6), 483–494.
About Dr. Sydney Ceruto
Dr. Sydney Ceruto is a Neuroscientist and Brain Performance Strategist specializing in neurological re-engineering for elite individuals navigating high-stakes environments. As the founder of MindLAB Neuroscience and the pioneer of Real-Time Neuroplasticity™, she translates clinical neurobiology into decisive competitive advantages for tech innovators, professional athletes, entertainers, and private families worldwide. Dr. Ceruto holds dual PhDs in Behavioral & Cognitive Neuroscience from New York University and dual Master’s degrees in Clinical Psychology and Business Psychology from Yale University. She is the author of The Dopamine Code, published by Simon & Schuster.
